ASTMH Annual Meeting 2025
blogRace to Treat a Brain-Eating Amoeba Reveals Lessons for Using Drug Screens to Locate Treatments for Rare Diseases
By: Matthew Davis, Burness
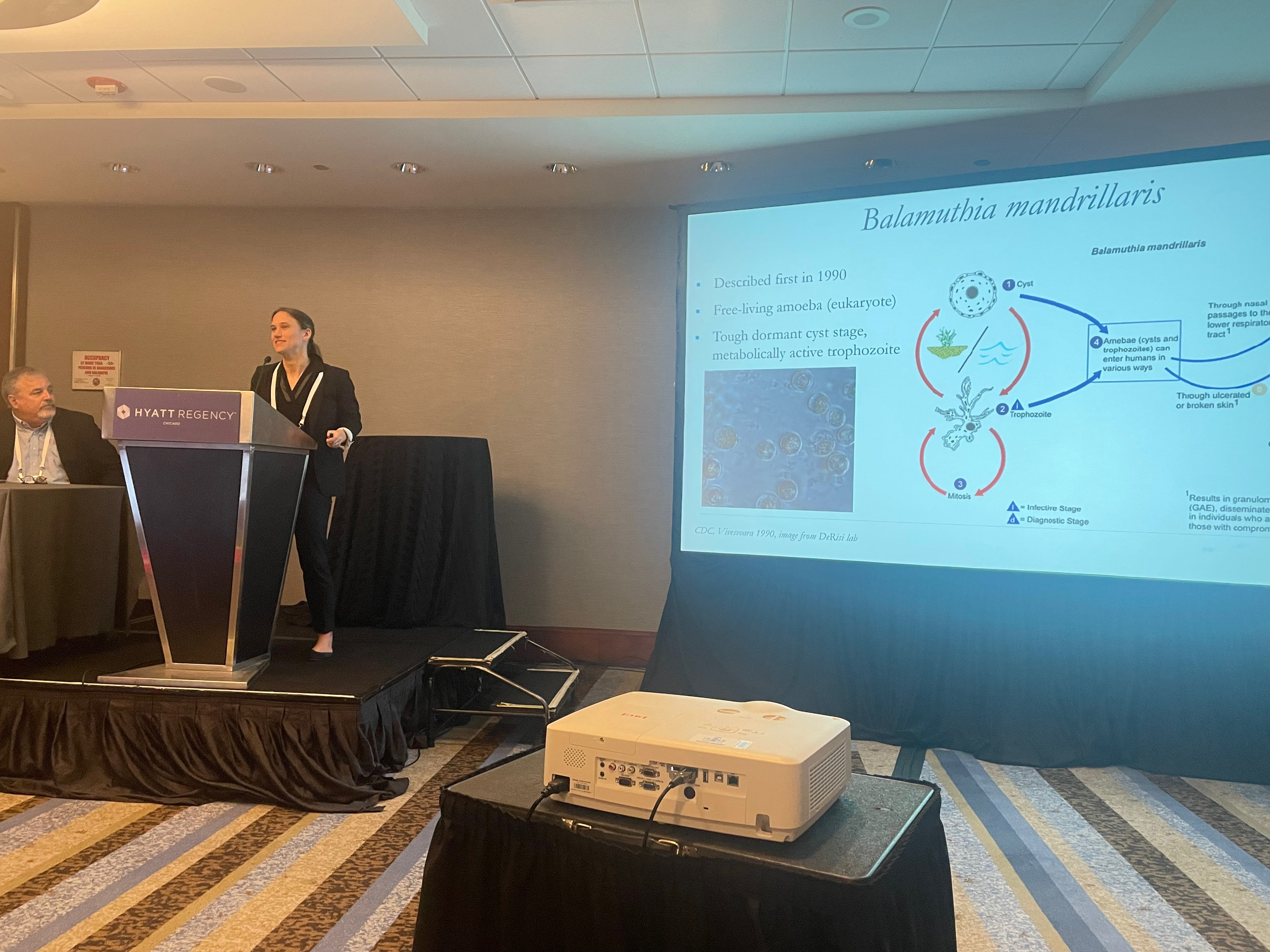
A researcher on Friday took her #TropMed23 audience on a journey to find a treatment for a rare disease that, if it was a movie, it would be part horror story, part thriller and ultimately a heart-warming story with a happy ending.
Natasha Spottiswoode, MD, PhD, with the University of California, San Francisco (UCSF), recounted her race to save a patient suffering from a rare and typically deadly “brain-eating” amoeba called Balamuthia mandrillaris. It eventually led her to a drug intended for something completely different — urinary tract infections — that proved to be highly effective. She said the experience offered lessons for others desperately seeking new therapies for rare diseases.
Spottiswoode said her quest began with a patient in his mid-50s who had suffered an unexpected seizure. Eventually, brain MRIs revealed lesions that led to a diagnosis of Balamuthia mandrillaris. It’s an extremely rare, extremely deadly disease. It’s caused by an amoeba that lives in soil and water and can make its way to the brain through contact with the skin, nose or mouth. Spottiswoode said that of the 123 documented cases in the literature, 90% were fatal.
A diagnosis of Balamuthia mandrillaris, she said, “is some of the worst news you can ever give to a patient.”
She noted that the only treatment regimen for the disease is complex, highly toxic and seldom successful. When she tried the recommended therapies for her patient, they caused renal failure and damage to his bone marrow.
Desperate for alternatives, Spottiswoode searched the scientific literature and discovered that in 2018, colleagues at UCSF had used a bioinformatics drug screening system to analyze more than 2,000 drug compounds to determine which one might be a good match for the pathogen. One clear winner had emerged. It was a drug called nitroxoline. It’s a type of antibiotic known as a quinoline that is routinely prescribed in Europe and China for urinary tract infections.
Problem solved!
Unfortunately, not quite, because nitroxoline was not approved for use in the United States. It ultimately took 41 days for Spottiswoode and her colleagues to navigate a maze of regulatory approvals and eventually obtain the drug from a supplier in China. As they worked the system, they managed to find a tolerable dosage of other medications that kept the patient alive. Spottiswoode said seven days after starting nitroxoline, a brain MRI showed “significant resolutions of the lesions” and the patient was “dramatically improved.”
Spottiswoode said she and her colleagues later wrote a case report and submitted it to a journal while also posting it to an online platform for non-peer reviewed studies, aka a “pre-print server.” That was fortuitous because it was picked up by a physician in 2022 searching for insights for treating a severely ill young girl in the Southeast who also had the disease. Spottiswoode said that as with her patient, nitroxoline led to a full recovery. The same thing happened just this summer, she said. The study was picked up by physicians in China treating a rapidly deteriorating patient who then recovered when put on a regimen of nitroxoline.
She said the experience taught her that while drug screens like the one that matched nitroxoline to the Balamuthia mandrillaris are no longer a novel idea, what is novel is finding a way apply them to patients--especially when if the screen identifies a drug not approved in your country.
“I learned that it’s essential to build partnerships with the FDA to make this a less challenging pathway,” Spottiswoode said.
She said the experience also taught her the value of pre-print servers.
“They allow you to quickly disseminate information to people who need it,” she said.
Spottiswoode said her research is now turning to understanding the biological mechanisms that have allowed a drug intended to treat urinary infection to be incredibly powerful for battling a brain-eating amoeba.
###
Related Posts
By: Matthew Davis, Burness